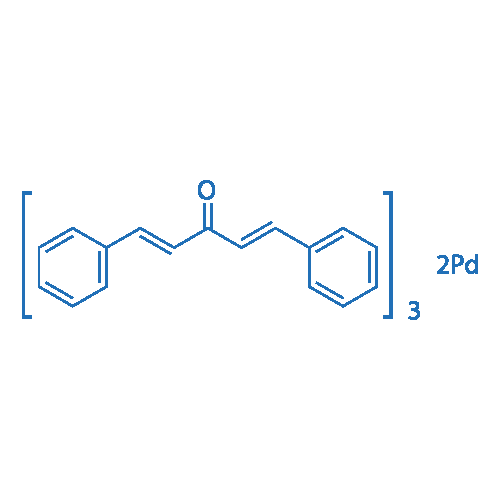
Degradation mechanism of Methyl Orange by electrochemical process on RuO(x)-PdO/Ti electrode. | Semantic Scholar
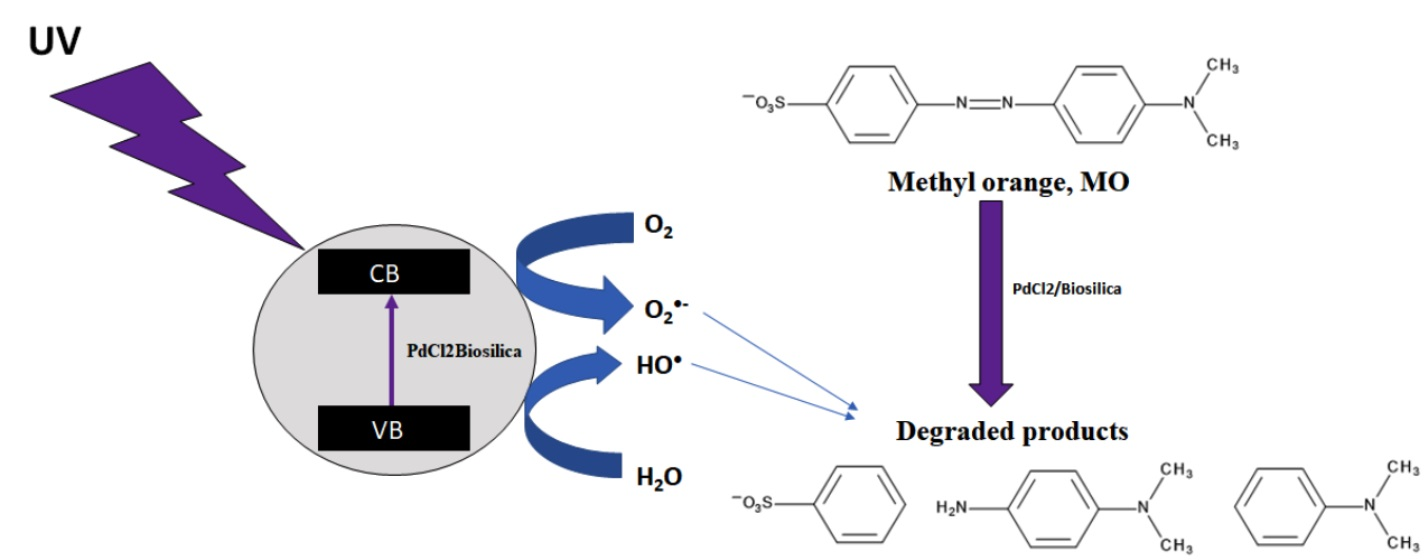
IJMS | Free Full-Text | Diatom Biosilica Doped with Palladium(II) Chloride Nanoparticles as New Efficient Photocatalysts for Methyl Orange Degradation
Tannic acid and palladium-modified magnetite nanoparticles for catalytic degradation of methyl orange - American Chemical Society

Palladium nanoparticles supported on ionic liquid and glucosamine-modified magnetic iron oxide as a catalyst in reduction reactions | SpringerLink

Polyaniline Supported Palladium Catalyzed Reductive Degradation of Dyes Under Mild Condition | Bentham Science
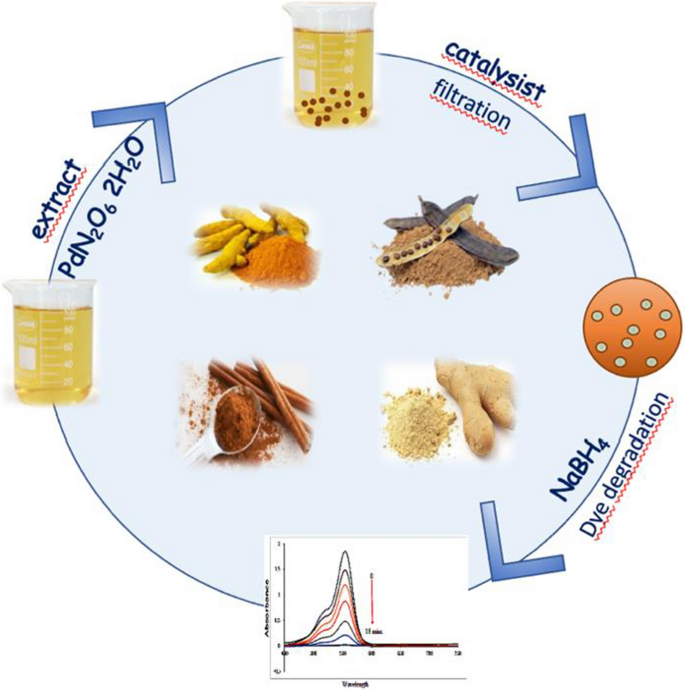
Green synthesis of palladium nanoparticles and investigation of their catalytic activity for methylene blue, methyl orange and rhodamine B degradation by sodium borohydride | SpringerLink

Green synthesis, characterization and catalytic degradation studies of gold nanoparticles against congo red and methyl orange - ScienceDirect

Figure 2 from Rapid degradation of azo dye methyl orange using hollow cobalt nanoparticles. | Semantic Scholar

Degradation mechanism and toxicity reduction of methyl orange dye by a newly isolated bacterium Pseudomonas aeruginosa MZ520730 - ScienceDirect

Enhanced Adsorptive Removal of Methyl Orange and Methylene Blue from Aqueous Solution by Alkali-Activated Multiwalled Carbon Nanotubes | ACS Applied Materials & Interfaces

Molecules | Free Full-Text | Synergistic Promotion of Photocatalytic Degradation of Methyl Orange by Fluorine- and Silicon-Doped TiO2/AC Composite Material

Degradation of methylene blue and methyl orange by palladium-doped TiO2 photocatalysis for water reuse: Efficiency and degradation pathways - ScienceDirect
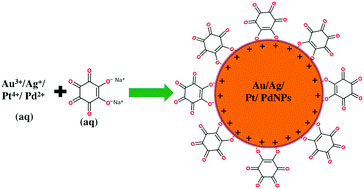
Green synthesis of gold, silver, platinum, and palladium nanoparticles reduced and stabilized by sodium rhodizonate and their catalytic reduction of 4-nitrophenol and methyl orange - New Journal of Chemistry (RSC Publishing)

Effective Catalytic Reduction of Methyl Orange Catalyzed by the Encapsulated Random Alloy Palladium‐Gold Nanoparticles Dendrimer. - Ilunga - 2017 - ChemistrySelect - Wiley Online Library

Acceleration of biotic decolorization and partial mineralization of methyl orange by a photo-assisted n-type semiconductor - ScienceDirect

Degradation mechanism of Methyl Orange by electrochemical process on RuO(x)-PdO/Ti electrode. | Semantic Scholar

Effective Catalytic Reduction of Methyl Orange Catalyzed by the Encapsulated Random Alloy Palladium‐Gold Nanoparticles Dendrimer. - Ilunga - 2017 - ChemistrySelect - Wiley Online Library

Pd–Rh Alloyed Nanoparticles on Zeolite Imidazolide Framework-67 for Methyl Orange Degradation | ACS Applied Nano Materials